By tailoring our solutions to address individual company needs and their priorities, we help our clients produce tangible results – from strategy through to your medical product or device launch. Through our network of resources and key technology partners, our services address the broad range of issues facing our clients in today’s ever-changing regulatory and technology-driven environment.
Warwick’s medical product and device design and commercialisation services enable you to bring your product to market quickly and efficiently.
We have been providing medical product design consulting services to the lifesciences Industry for twenty-five years. Our development offerings draw upon our broad capabilities in the following areas: early day concept investigations, technical development, user engineering, technology strategy, commercialisation, and monitoring and improvement.
We can drive the design with innovative concept prototypes to demonstrate clinical potential, detail the concept for a manufacture, transfer the design to production of the full medical product or device – on time and within budget.
At this industrial design consultants we undertake proof of principle models, development of patient compliance devices and technical and engineering design. We design for manufacturability, and follow it up with design verification and process validation then transfer to production.
Design and development is seen through handling and human factors engineering, tolerancing, metrology to industrialisation and scale-up working closely with toolmakers, moulders and assemblers.
In our laboratory we have Class 10 clean assembly areas and fume cabinets for using active or hazardous material in developments and test.
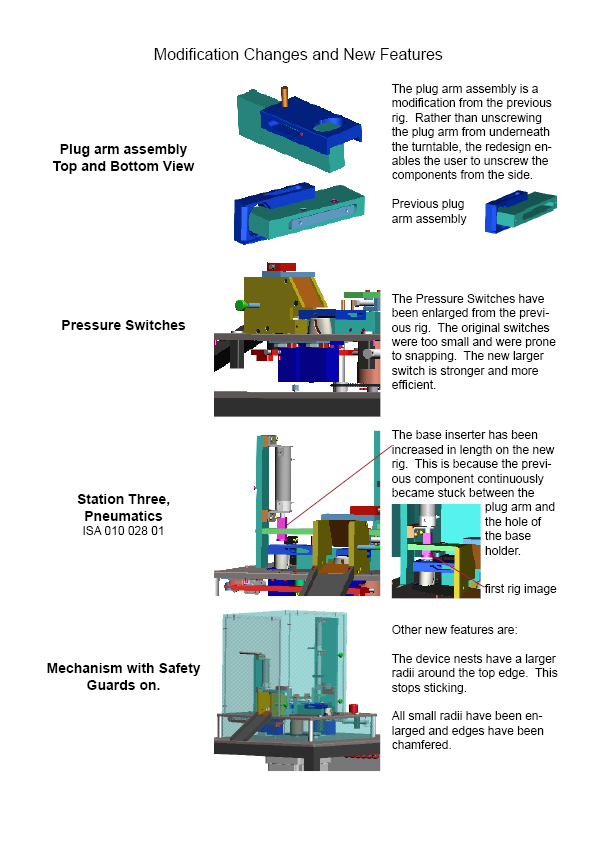
To support the products we design we also design and manufacture laboratory and production line test, evaluation and assembly machines and devices.
Specialist lift bathing
Home Dialysis
Manufacturing self injector test machines
Drug lock boxes
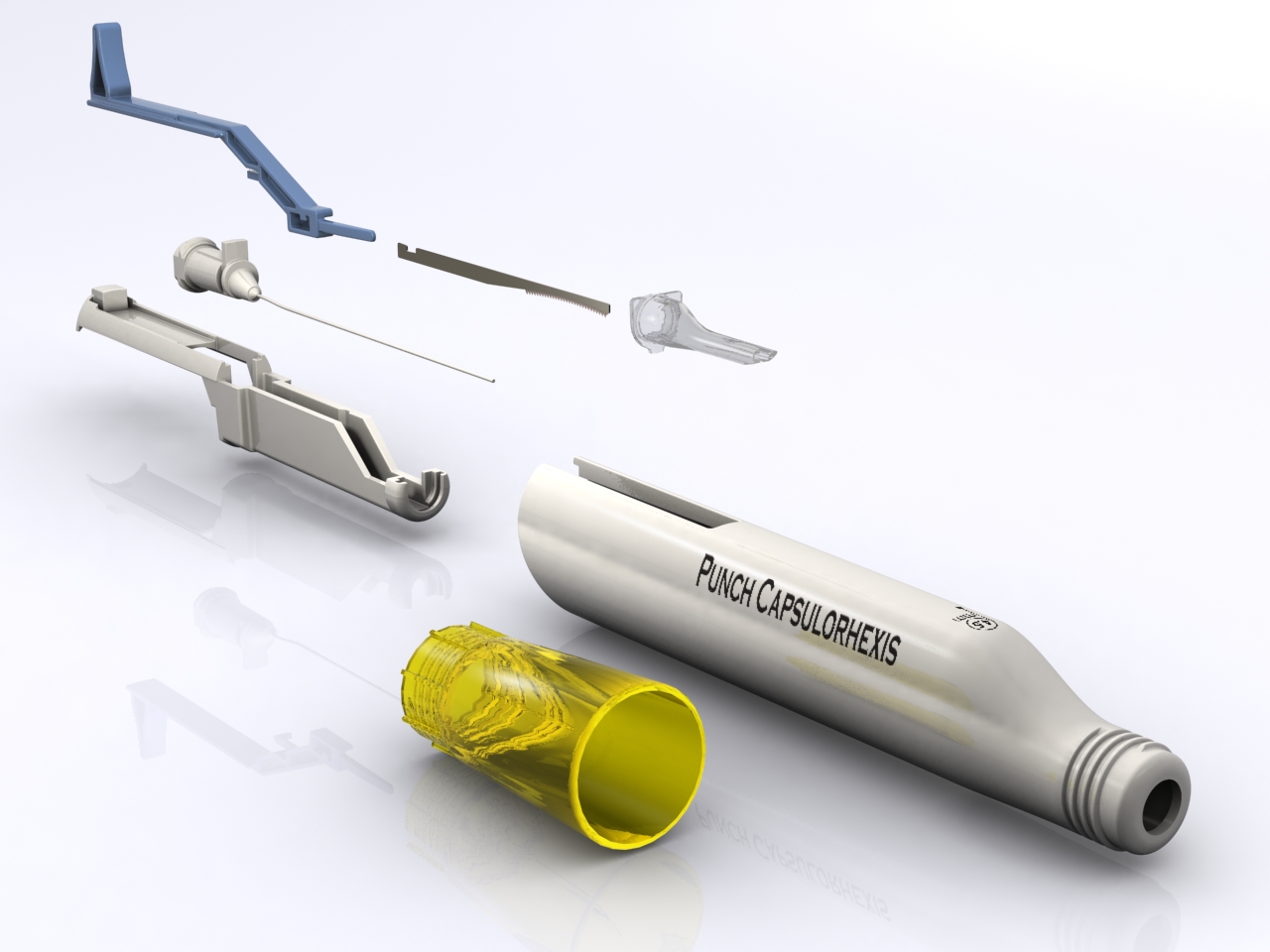
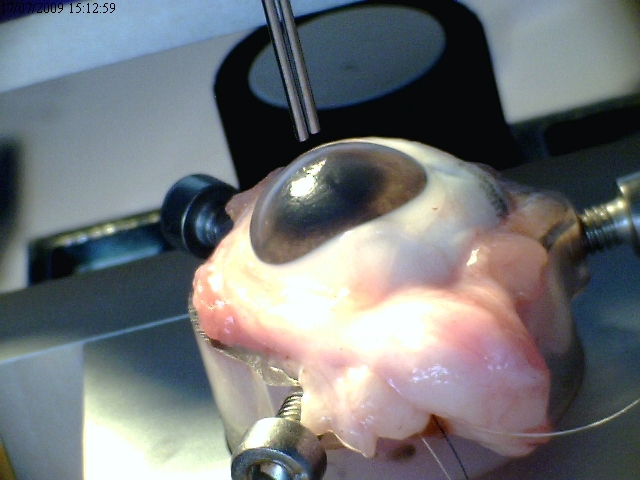
Ophthalmic surgery
Theatre equipment appearance
Dermatological incision
Automated reconstitution device
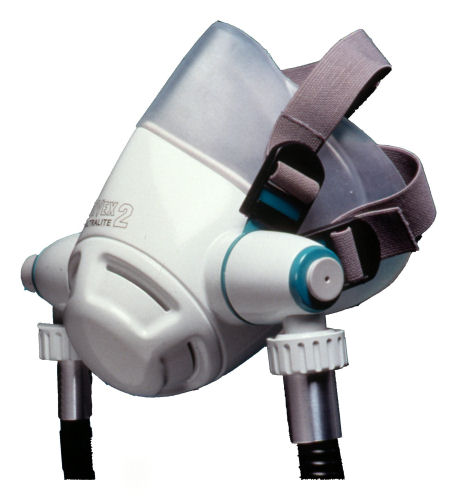
Hyperbaric chamber breathing equipment
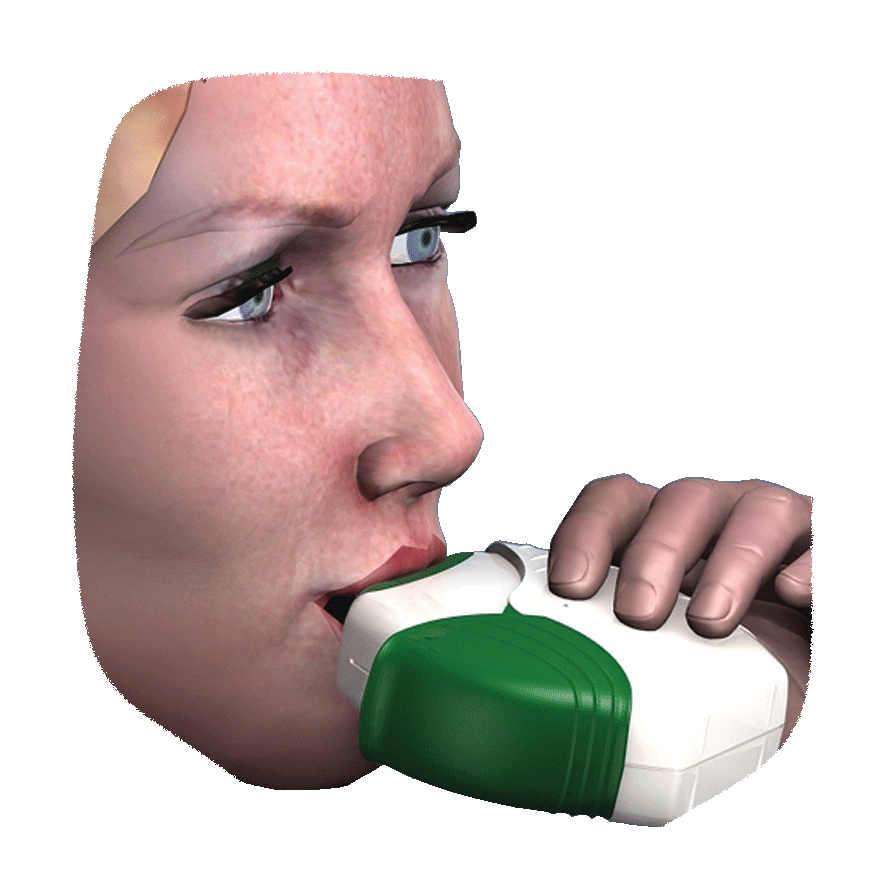
Inhalers
Inhaler training devices
Demonstration machines
Physiotherapy devices
Healthcare hygiene tracking
Artificial organ growth support devices
Novel tablet and capsule dispensing consumer packaging